OriGro™ Active
Cellular Bone Matrix
A next-generation cellular bone matrix engineered to deliver the biologic performance of autograft.

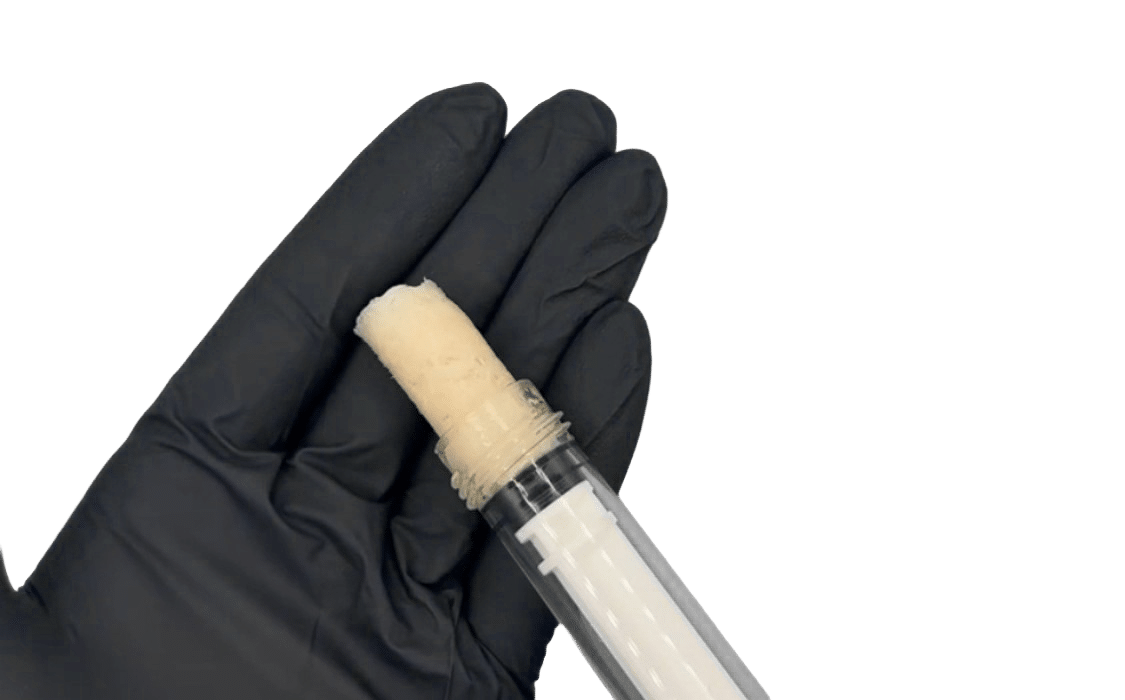
Built for Surgical Control. Designed for Fusion Confidence.
- Exceptional handling characteristics for
precise graft placement - DMSO-free preservation technology
maintaining viable cell integrity - Zero rinsing required
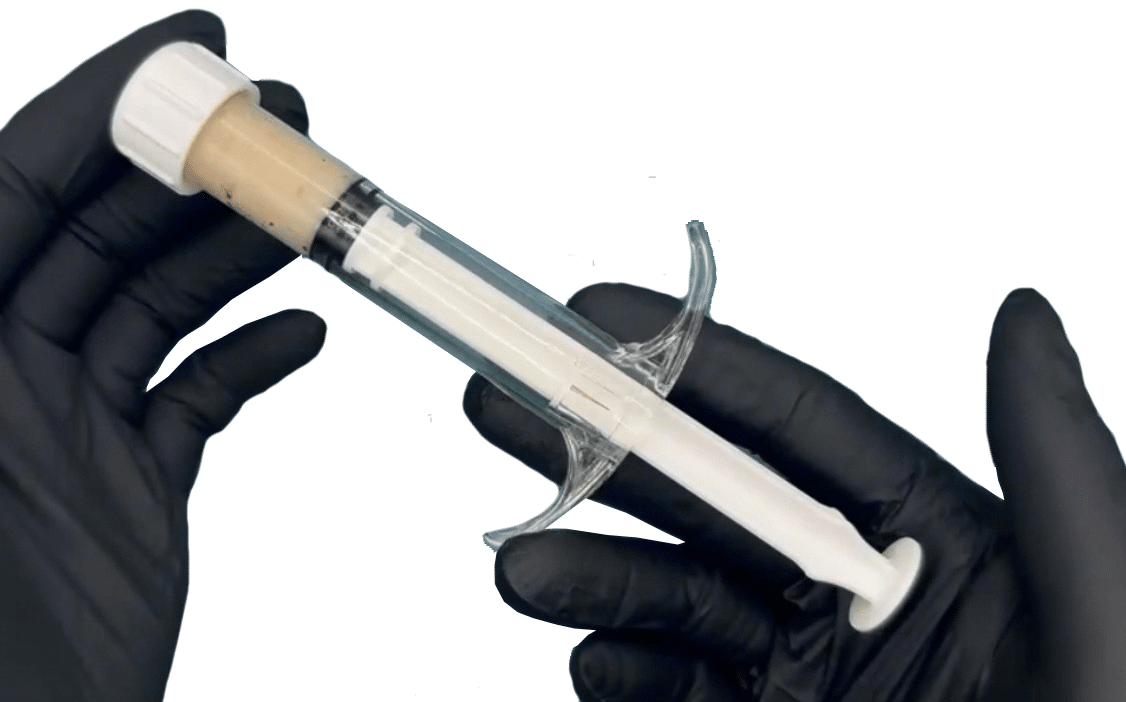
- Syringe delivery for surgical efficiency
- 10-minute thaw time
- 4-hour post-thaw usability window
- Non-immunogenic composition
- Rigorous donor screening and
biocompatibility assurance
System Features
By integrating a structurally optimized scaffold, preserved osteoinductive signaling, and a high concentration of viable osteogenic cells, OriGro Active provides advanced bone regeneration in a surgically ready format with exceptional handling characteristics.

Osteoconductive Bone Scaffold
A composite of cancellous mineralized bone matrix and demineralized cortical fibers creates a structurally optimized framework for cellular attachment and migration, promoting rapid integration at the fusion site.1,2

Osteoinductive Growth Factors
OriGro’s demineralized cortical fibers preserve a native cascade of growth factors stimulating the recruitment of cells needed for bone healing. Validated in vivo osteoinductive potential supports biologic performance in challenging healing environments.2

High-Concentration Osteogenic Cells
Angiogenic Activation
Specifications
Size Options
1 cc
2.5 cc
5 cc
10 cc
15 cc
Resources
Brochures
Additional Information
Contact Us
USA: 800 447-3625
To submit a complaint, please email SpineComplaints@highridgemedical.com
10225 Westmoor Dr. Westminster, CO 80021 USA
To obtain a copy of the current Instructions for Use (IFU) for full prescribing and risk information, please call 800 447-3625.
References
- Martin GJ, Boden SD, Titus L, et al. Spine 1999.
- Data on file at Origin Biologics.
- Marsell R, Einhorn TA. Journal of Bone and Joint Surgery 2011.
Legal Manufacturer
Origin Biologics, LLC
6635 S Eastern Ave #100
Las Vegas, NV 89119 USA